Introduction
The Board of Medicine meets six times each year to conduct disciplinary hearings, licensure hearings and to handle board business including creating and amending rules of the Board. At this meeting, the Board recognized Nabil El Sanadi, M.D. for serving as Chairman in 2014. Dr. El Sanadi spearheaded discussions concerning the Board’s telemedicine and office surgery rules. The Board also recognized two medical students: Kyle R. Diamond from Florida Atlantic University, and Jason I. Liounakos from Florida International University. These medical students were honored because they exemplify the professionalism, moral character, compassion and intellect essential to future leaders of the medical profession. Dr. Orr also instituted the Resident Recognition Program whereby the Board will recognize current residents in an approved ACGME program. Contact the Board office at (850) 245-4132 for nomination criteria.
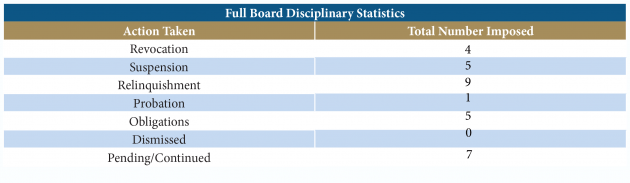
Full Board Disciplinary Statistics
At the February meeting, the Board held 31 disciplinary hearings, which resulted in disciplinary action as outlined in the chart below. The Board recognizes that the overwhelming majority of Florida’s licensees will never be engaged in a disciplinary proceeding. However, in the cases that come before the Board in which there is an alleged violation of Florida’s Medical Practice Act, the members are committed to the fair and just application of the law to protect the citizens and visitors of Florida. The disciplinary hearing agenda is available on the Boards’s website at www.FLBoardofMedicine.gov. All Final Orders regarding these disciplinary hearings are available online at www.FLHealthSource.gov by clicking on Verify a License and searching the licensee’s name.
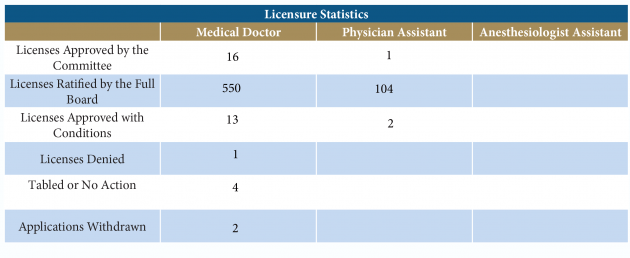
Licensure Statistics
The statutory scheme of licensing medical doctors in Florida provides for various avenues of eligibility. The licensing of these physicians includes an extensive background investigation on each applicant, which includes 8-10 years of medical education, postgraduate training, and for some applicants, extensive practice experience. The process requires the evaluation of the applicant’s medical credentials, and all information, reports, and documents obtained in the application review process to determine the applicant’s eligibility for licensure. Applications that contain any conflicting information or questionable information are submitted to the Credentials Committee for review.
The Credentials Committee presents application recommendations (approval or denial) to the Board at the Board’s regular bimonthly meetings. Applicants with no questionable application materials are issued a license and the licenses are ratified by the Board at the next meeting. Over the past year, the Board has worked to make licensure in Florida a more efficient process. The application has been revised and is available at https://flboardofmedicine.gov/licensing/.
Rule Discussions
The Board held a hearing concerning Rule 64B8-10.003, F.A.C. – Costs of Reproducing Medical Records and approved draft language. In addition, the Board heard testimony regarding Rule 64B8-9.009, F.A.C. – Standards of Care for Office Surgery and approved draft language for that rule. When the changes become effective, we will make an announcement on the Board’s web page at www.FLBoardofMedicine.gov. Please stay tuned for more information.

February 2015 Florida Board of Medicine Meeting Review (pdf)